Professor XU Aimin

Rosie T T Young Professor in Endocrinology and Metabolism
Professor, Chair of Metabolic Medicine
Director of State Key Laboratory of Pharmaceutical Biotechnology
- BMed (Anhui), MSc, PhD (Auck)
| amxu@hku.hk | |
| 2819-9754 | |
| HKU Scholar Hub |
Biography
Research Profile
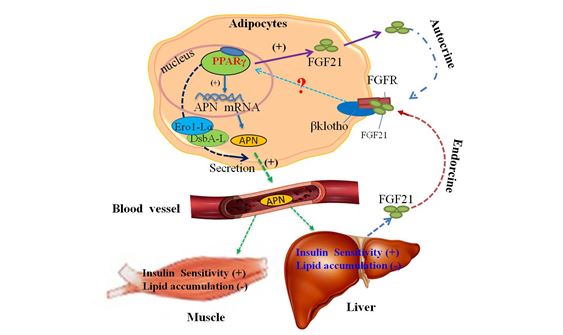
- Identification and functional characterization of adipokine netowork: Using an integrated proteomics-based approach, we have identified multiple posttranslationally modified isoforms of adiponectin, and have demonstrated that hydroxylation and subsequent glycosylation of several lysine residues at the collagenous domain are obligatory for insulin-sensitizing effects of adiponectin by promoting the formation of high molecular weight oligomeric complexes (Wang Y, JBC, 2002, 2005, 2006; Biohcem. J, 2008; Xu A, JCI, 2003). We have also identified the circulating form of adipocyte fatty acid-binding protein (A-FABP) and lipocalin-2 as pro-inflammatory adipokines mediating toxic lipids-induced insulin resistance and vascular dysfunction (Xu A, Clin Chem: 2006; Circulation: 2007; Hui X, JBC:2010). More recently, we have demonstrated that fibroblast growth factor (FGF21), an anti-diabetic hormone traditionally thought to be produced from hepatocytes, is also secreted from adipose tissues (Zhang X, Diabetes, 2008 and Lin Z, Cell Metabolism, 2013). Furthermore, we found that adipose-derived FGF21 exerts its anti-diabetic activities by inducing adiponectin production (Fig 1). Currently, we are investigating the mechanisms by which FGF21 controls adiponectin production under physiological and pathological conditions.
Fig 1. Adlponectin confers the metabolic actions of FGF21 in the liver and skeletal muscle
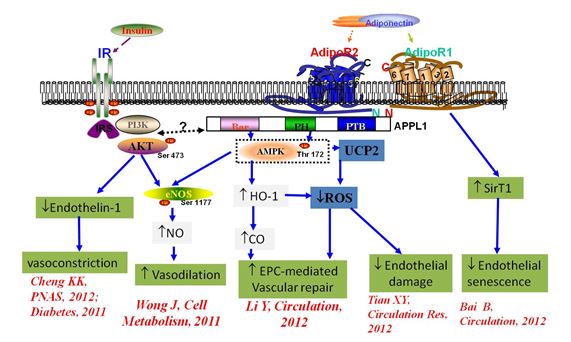
2. Signaling mechanisms underlying the insulin-sensitizing and vascular protective effects of adiponectin: Adiponectin is one of the few adipokines with insulin-sensitizing and cardiovascular protective properties, but its mechanisms of actions remain poorly characterized. In 2007, We identified APPL1, an intracellular adaptor protein, as a key signaling molecule that bind to adiponectin receptors mediating adiponectin-induced activation of AMP-activated protein kinase (AMPK), thereby inducing eNOS activation and NO production in endothelial cells (Cheng KK, Diabetes, 2007). Our subsequent studies demonstrated that APPL1 is also a key component of insulin-evoked signaling cascade, by promoting membrane targeting and translocation of the protein kinase Akt (Cheng KK, Cell Metab. 2009; BJ, 2013). Furthermore, using APPL1 transgenic and knockout mice, we have found that APPL1 plays a central role in controlling vascular homeostasis as well as insulin secretion (Cheng KK, PNAS, 2012; Wang Y, Diabetes, 2011).
We have also uncovered an important role of adiponectin in preventing diabetes-induced vascular injury, by promoting the functions of endothelial progenitor cells (EPCs) via an AMPK-dependent mechanism (Chang J, Diabetes, 2010). In light of the key role of AMPK in mediating the endothelium-protective effects of adiponectin, we have generated transgenic mice with endothelium-selective constitutive AMPK activation, and found that endothelium-selective activation of AMPK is sufficient to prevent diabetes-associated impairment of vascular repair via induction of heme oxygenase-1 (HO1) pathway, which in turn augments EPCs-mediated endothelial regeneration (Li Y, Circulation, 2012) (Fig 2). We are currently investigating the roles of FGF21/adiponectin axis in protecting obesity-induced vascular inflammation and atherosclerosis in both rodents and large animals.
Fig 2. Multiple signaling pathways contribute to the vascular protective effects of adiponectin
Selected Publications
- Lin Z, Tian H, Lam KS, Lin S, Hoo RC, Konishi M, Itoh N, Wang Y, Bornstein SR, Xu A*, Li X. Adiponectin mediates the metabolic effects of FGF21 on glucose homeostasis and insulin sensitivity in mice. Cell Metabolism. 2013, 17:779-89 (cited ~30 times within 8 months after publication)
- Cheng KK, Lam KS, Wu D, Wang Y and Xu A*. APPL1 potentiates insulin secretion in pancreatic beta-cells by increasing Akt-dependent expression of SNARE proteins in mice.Proc. Natl. Acad. Sci. USA. 2012, 109:8919-24, selected for commentary.
- Li FY, Lam KS, Tse HF, Chen C, Wang Y, Vanhoutte PM, Xu A*. Endothelium-selective activation of AMP-activated protein kinase prevents diabetes mellitus-induced impairment in vascular function and reendothelialization via induction of heme oxygenase-1 in mice. Circulation. 2012, 126:1267-77.
- Ye D, Li Y, Lam KS, Li H, Jia W, Wang Y, Man K, Li X and Xu A*. TLR4 mediates obesity-induced nonalcoholic steatohepatitis through activation of X-box binding protein in mice. Gut. 2012, 61: 1058-67
- Wang Y, Cheng KK, Lam KS, Wu D, Wang Y, Huang Y, Vanhoutte PM, Sweeney G, Li Y, Xu A*. APPL1 counteracts obesity-induced vascular insulin resistance and endothelial dysfunction by modulating the endothelial production of nitric oxide and endothelin-1 in mice. Diabetes, 2011, 60: 3044-54.
- Wong WT, Tian XY, Xu A*, Yu J, Lau CW, Hoo R, Wang Y, Lee VW, Lam KS, Vanhoutte PM, and Huang Y. The obligatory role of adiponectin in restoring endothelial function in PPARγ agonist-treated diabetic mice. Cell Metabolism, 2011, 16:101-15.
- Chang J , Li Y , Huang Y , Lam KS, Hoo LC, Wong WT, Cheng KK, Wang Y, Vanhoutte PM , and Xu A*. Adiponectin Prevents Diabetic Premature Senescence of Endothelial Progenitor Cells and Promotes Endothelial Repair by Suppressing the p38 MAP kinase/p16INK4A Signaling Pathway. Diabetes, 2010, 59: 2949-59
- Cheng KK, Iglesias MA, Lam KS, Wang Y, Sweeney, Zhu W, Vanhoutte PM, Kraegen EW and Xu A*. APPL1 Potentiates Insulin-mediated Inhibition of Hepatic Glucose Production and Alleviates Diabetes via Akt Activation in Mice. Cell Metabolism, 2009, 9:417-27.
- Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou ZG, Liu F, Wong RL, Chow WS, Tso AW, Lam KS, Xu A*. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes, 2008, 57:1246-53.
- Cheng KK, Lam KS, Wang Y, Huang Y, Carling D, Wu D, Wong C, Xu A*. Adiponectin-induced endothelial nitric oxide synthase activation and nitric oxide production are mediated by APPL1 in endothelial cells. Diabetes. 2007, 56:1387-94 (cited over 170 times).
- Xu A*, Lam MCL, Chan KW, Wang Y, Hoo RCL, Zhang JL, Tso A and Lam KSL, ANGPTL4 decreases blood glucose, improve glucose tolerance, but induces hyperlipidemia and hepatic steatosis in mice, Proc. Natl. Acad. Sci. USA, 2005, 102, 6089-6091
- Xu A*, Wang Y, Keshaw H, Xu LY, Lam KS, Cooper GJ. The fat-derived hormone adiponectin alleviates alcoholic and nonalcoholic fatty liver diseases in mice. J. Clin. Invest. 2003, 112:91-100. (selected for the cover story of this issue, CITED OVER 980 times).
Key Offices
- Director, State Key Laboratory of Pharmaceutical Biotechnology, HKU
- Director, Antibody and Immunoassay Services, HKU
- Deputy Chair, Biochemical Journal ( from June 2013-)
- Editor, Clinical Science (from 2008-)
- Editorial Board member, Journal of Obesity (from 2008-
- Editorial Board member, Journal of Diabetes (from 2009-)
- Academic Editor, PLOS One (from 2007-)
- Editorial board member, Adipocytes (from 2011-)
- Editorial board member, Cardiovascular Drugs and Therapy (from 2010-)
Patents
- Lipocalin-2 antibodies for methods of treatments. US patent number: 8,481,032B2
- Lipocalin-2 as a prognostic and diagnostic marker for heart and stroke risks, US patent number: 8,030,097 B2
- Phosphoprotein target for insulin and insulin antagonists. (US patent number: US 6,884,575)
- Adiponectin and uses thereof. (US patent Number: 7,365,170)
- Lipocalin-2 as a diagnostic marker and therapeutic target. US patent No: 7,645,616.
Applied research
- Strategic alliance with Servier (a European-based multi-national pharmaceutical company) to jointly develop drug candidates by targeting adipokines (~ 5 MILLION HKD);
- Development and commercialization of over 20 immunoassay kits for research and clinical diagnosis of diabetes and cardiovascular disorders.